Certified Professional Medical Products CPUX-F ENGLISH UA3013 Singapore
- The seminar teaches all important concepts for the development and design of medical devices according to EN62366 / IEC 62336. From the basics of ISO 9241, to use context analysis, persona, conception, prototyping up to the usability testing + test + certificate
- Globally Recognized Certifications: The UXQB® Certified Professional for Usability and User Experience - Foundation Level (CPUX-F) certifies that the certificate holder is familiar with the basic concepts and terminology of the most important areas of expertise of usability and user experience
- Unique: Planning, execution and evaluation of usability evaluation with real users in the seminar and remotely / unmoderated usability tests with real users and video recording.
- Proximity to practice:The focus of the seminar is on practical exercises. There are extensive practical exercises in the areas of touch design, tablet and mobile for medical devices for all concepts of the CPUX seminar parts.
- UXQB®: The course teaches the most important Usability & UX methods according to the official criteria and syllabus of the UXQB (Usability & UX Qualification Board).
- Approved UXQB® Certificate: Certified Professional for Usability and User Experience - Foundation Level (CPUX-F). The "UXQB® Certified Professional for Usability and User Experience - Foundation Level (CPUX-F)" certificate certifies that the certificate holder is familiar with the basic terms and concepts of the most important areas of expertise of usability and user experience.
- UX Training according to International Usability and User Experience Board (UXQB) inkl. exercises
- Exam preparation training for „UXQB® CPUX-F certification
- LIVE ONLINE Training with V-Classroom-XL - Video Audio - ONLINE REALTIME WHITEBOARD - eLearning Portal - Virtual Classroom
- 5 modules a 4 hours live online V-Classroom-XL training & workshop + test + certificate
- 2 modules per week thuesday and thursday, last week only thuesday
- Singapore 02:00 pm - 06:00 pm local time
- Tel Aviv 09:00 am - 01:00 pm local time
- Bengaluru 11:30 am - 03:30 pm local time
- Dubai 10:00 am - 02:00 pm local time
- Berlin 08:00 am - 12:00 am local time
- 20 hours full of UX methods, concepts
- 10 Participants maximum - NO mass training - much time for individual questions
Duration:
5 modules
Optional: CPUX-F exam (Please mention upon registration if you would like to opt for the exam.)*
Target Group:
- Manufacturer of medical devices
- Product Manager medical devices
- User Interface Designer
- Software Engineer
- Project Manager
- Quality Manager
- All those who are involved in development projects who collaborate and influence the design of medical devices
- Marketing
Seminar Content:
CPUX-F Certification exam: 300,- plus VAT
Seminar including examination & certificate of Usability Academy
Event Information
| Event Date | 22.09.2026 14:00 |
| Event End Date | 06.10.2026 18:00 |
| Capacity | 12 |
| Original Price | 1,195.00€ |
| Discounted Price | 1,075.50€ (Until Date 21.07.2026 0:00 ) |
| Location | Singapore |
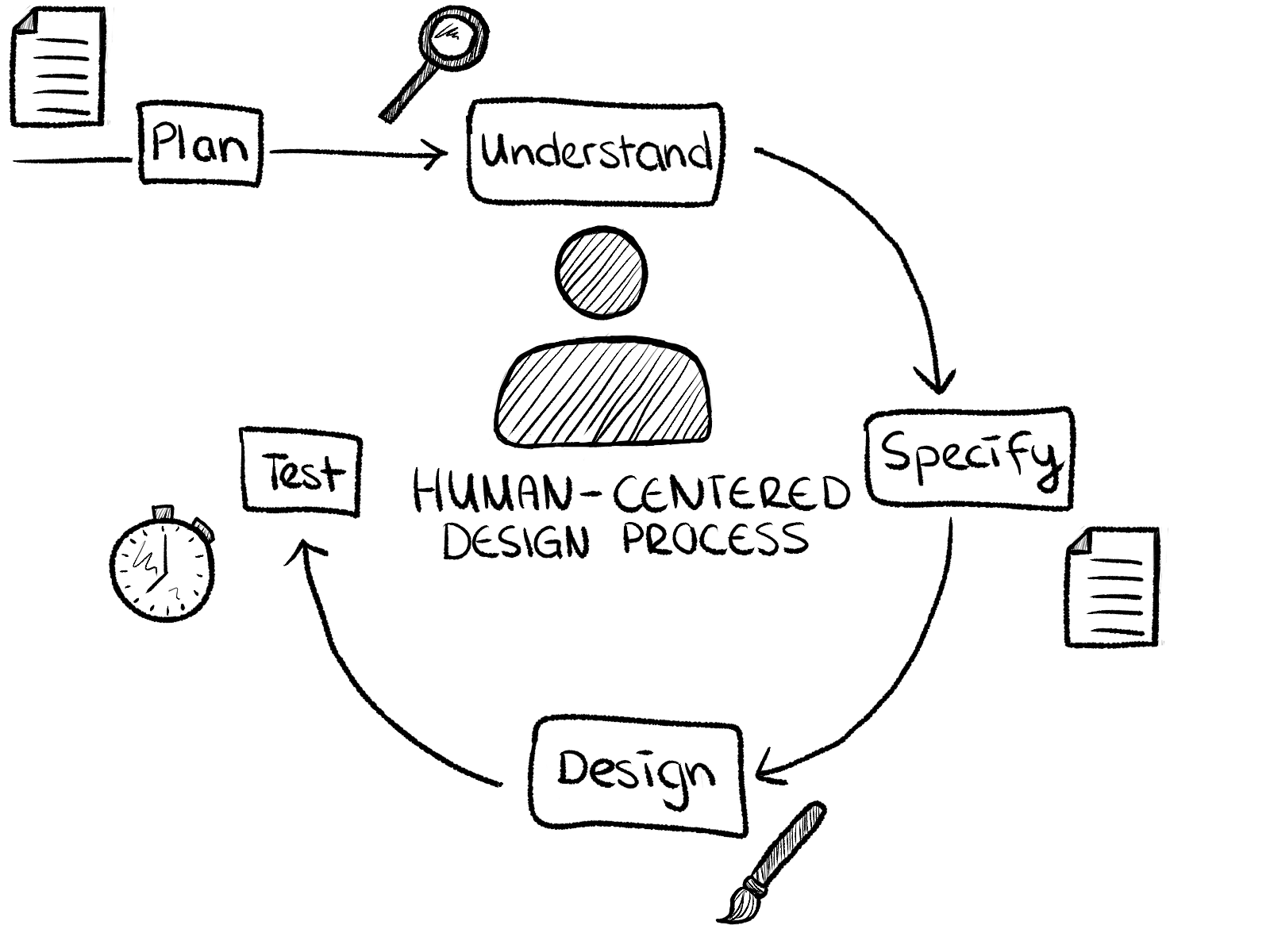 01. human-centred design process according to ISO 9241-210:2010
01. human-centred design process according to ISO 9241-210:2010